Ph, WHat is it?
Ph is the measure of the molar concentration of Hydrogen ions in an aqueous substance. The formula in calculating the Ph of a substance is Ph = -log[H]. Where -log refers to the logarithmic scale and [H] refers to the Hydrogen or Hydronium ion concentration in an aqueous solution in moles per litre.
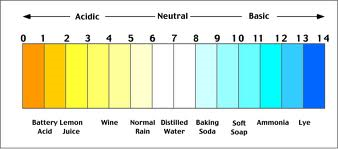
Acids and Bases are categorized through the Ph scale. An Acid is a Ph lower than 7 and a strong acid is a Ph of 1-2. A base on the other hand, is where the Ph is higher than 7 and a strong base is a Ph of 13-14. A neutral substance, has a Ph of 7. An example of this, is distilled water
Acids are sour in taste and display a red colour when tested on litmus paper. It reacts with a base (neutralization reaction) to form a product of salt and water. A base is a bitter tasting substance and displays a blue colour on litmus paper.
Acids and Bases are categorized through the Ph scale. An Acid is a Ph lower than 7 and a strong acid is a Ph of 1-2. A base on the other hand, is where the Ph is higher than 7 and a strong base is a Ph of 13-14. A neutral substance, has a Ph of 7. An example of this, is distilled water
Acids are sour in taste and display a red colour when tested on litmus paper. It reacts with a base (neutralization reaction) to form a product of salt and water. A base is a bitter tasting substance and displays a blue colour on litmus paper.